What are Post-Translational Modifications?Post-translational modifications (PTM) are chemical changes made to a protein after it has been translated from a mRNA sequence into a protein sequence. These chemical modifications play a large role in the overall function of proteins, including protein activation, transport, and degradation [1]. Modifications can be either reversible or irreversible. Reversible modifications are chemical changes which involve the addition of chemical groups, complex molecules, and polypeptides, while irreversible modifications involve the cleavage of peptides and amino acid modifications (Figure 1) . Specifically, Phosphorylation is an important, reversible PTM which involves the addition of a phosphate group to a specific amino acid residue, typically serine, threonine, or tyrosine. Phosphorylation or dephosphorylation, the cleavage of a phosphate group, can result in either the activation or deactivation of many proteins, leading to many changes within a cell.
|
Figure 1: Types of reversible and irreversible protein modifications.
|
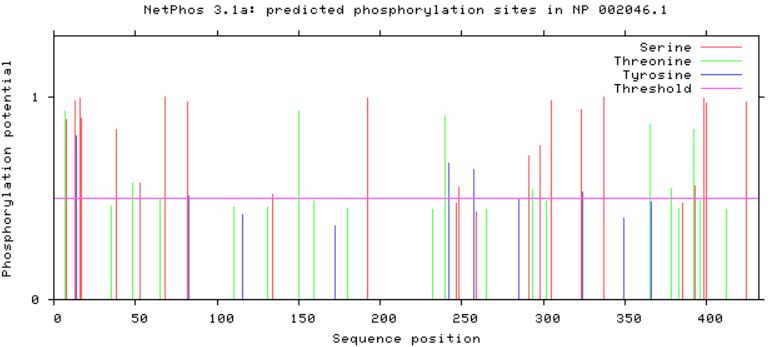
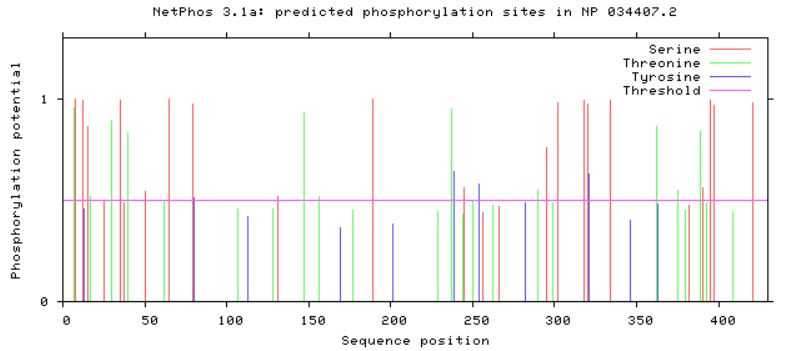
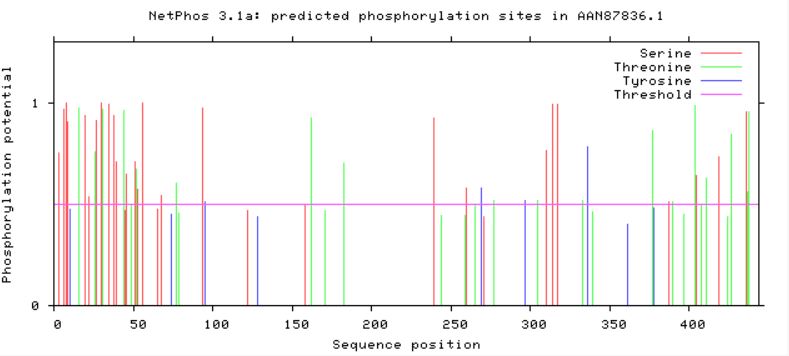
Phosphorylation Sites on GFAPUtilizing the publicly available website, NetPhos, a protein sequence of interest can be uploaded and analyzed in order to find predicted phosphorylation sites. Serine, threonine, and tyrosine phorphorylation sites were predicted for GFAP in humans, as well as in mouse and zebrafish homologs (Figures 2,3 and 4). Conserved residues which could be phosphorylated were found between all three homologs, with mouse and humans sharing more conserved sites than humans and zebrafish (Figures 2,3, and 4).
|
Discussion
Looking at predicted phosphorylation sites in GFAP homologs can provide new information regarding which sites are crucial to protein function based on site conservation. It is important to keep in mind that when looking at zebrafish predictions compared to human and mice, zebrafish have a longer GFAP protein sequence, and their conserved residues can be found approximately 50 amino acids downstream. Accounting for the longer zebrafish GFAP homolog, conserved sites were found among human, mouse, and zebrafish GFAP. These residues can serve as new potential targets for phosphorylation modification in future studies in order to reveal more information about how GFAP functions in neuronal upkeep and myelination, along with other important processes GFAP plays a role in.
References
[1] Bürkle, A. (2001). Posttranslational Modification. In S. Brenner & J. H. Miller (Eds.), Encyclopedia of Genetics (p. 1533). New York: Academic Press. https://doi.org/10.1006/rwgn.2001.1022
[2] NetPhos 3.1 Server. Retrieved April 11, 2018, from http://www.cbs.dtu.dk/services/NetPhos/
[3] Skop, A. (2018). Lab 8: Proteomics. Retrieved April 12, 2018, from http://genetics564.weebly.com/proteomics-i.html
[2] NetPhos 3.1 Server. Retrieved April 11, 2018, from http://www.cbs.dtu.dk/services/NetPhos/
[3] Skop, A. (2018). Lab 8: Proteomics. Retrieved April 12, 2018, from http://genetics564.weebly.com/proteomics-i.html